What would you do if you were faced with chronic, debilitating pain every day? Never knowing when you could possibly be hit with a wave of cramps so severe that you feel paralyzed? If you were constantly fatigued, so much so that sleeping for days would not give you energy?
Would you take a pill that promised to reduce some of that pain? Something that your friends with the same symptoms were calling a 'miracle drug'? Would you take it even if it forced you into early menopause, made your bones thin and could give you suicidal thoughts?
This is the dilemma that women with endometriosis in America are currently facing, as a new treatment, Orilissa, has come into the market. After ten years without one single new treatment, Orilissa was FDA-approved yesterday, and has been dubbed 'a significant advancement' by Michael Severino, M.D, also the executive vice president of AbbVie - the drug company producing the pill.
Tested on 1700 women across two studies, the treatment reduced pain during menstruation, non-menstrual pelvic pain, and pain during intercourse, according to the press release. It's been dubbed a 'miracle drug' within the endometriosis community, however, it also forces your body into early menopause, according to the U.S Department of Health and Human Services.
Pause: Forced early menopause? Really? That’s the dream treatment everyone is excited about? Not only does it put you into the early process of hot flashes, night sweats, mood changes and beyond, the most serious side effect is actually a decrease in bone mineral density, so your bones will become thinner. According to AbbVie, there is also a risk of suicidal thoughts, actions or behaviour.
We weren't the only ones who hesitated when reading the side-effects, Neelam Heera, who suffers with endometriosis herself, was also taken aback. ‘I don’t think it's as all singing and dancing as it wants to be,’ says Neelam Heera, who suffers with endometriosis herself, ‘It's almost put out there as a miracle drug but really the side effects are just as bad.’
As the founder of Cysters, a self-help group for women who suffer with reproductive health issues, Neelam has seen first-hand the desperation women are led to in finding a suitable treatment for endometriosis. ‘I think the difficulty is, because you're in so much pain and there isn't enough accessible information out there, you've got the problem of very desperate, vulnerable women clinging onto hope and then just going for it’, she continued, ‘without necessarily understanding what it’s really going to do to them’
While Orilissa isn’t available in the UK just yet, given the severe side-effects, Neelam fears what would happen if it was. ‘I suspect if it was in the UK people would just rush to try it, purely for the fact it's been put out there like a miracle drug’.
The drug being seen as a miracle is exactly what Emma Cox, chief executive of Endometriosis UK, wants to avoid. ‘It’s not a cure for the disease,’ she said, ‘and I think we need to be clear with women so we don’t get peoples hopes up that they can suddenly take a drug and that’s it.’
That being said, Emma explains that seeing a new treatment for endometriosis is positive, and that despite the concerns around side-effects, thinks Orilissa should be made available in the UK so women at least have more choices in their treatment.
‘I would like to see it available in the UK as an option, as there’s currently no specific drugs designed just for endometriosis available in the UK,’ she continued, ‘1.5 million women have the condition, it has a huge potential impact on women’s lives and should [Orilissa] help some women, they should be given that choice’
Whether or not it will be made available in the UK is the real question, and from what Emma says, it doesn’t sound likely. ‘There is another drug for endometriosis in Europe and America called Visanne that isn’t available in the UK on the NHS,’ she said, ‘so we haven’t even got the previous drug available.’
However, as a charity, Endometriosis UK would like this to change, making Visanne and Orilissa available to women in the UK, but also increasing research into the condition to help people understand it better and allow for more treatments to be created.
‘We’d like to see the government making available the drugs that are available in other countries,’ she continued, ‘[Orilissa] isn’t licensed in the UK so no one here has it even as an option to see if it would be good for them. This isn’t a rare disease, its 10% of women, and we really do need to start getting ways of recognising the condition, diagnosing it and then helping women live with it, it would be great to see more drugs coming through’
For too long we have been subject to this same cycle, new treatments for women’s health issues are developed, everyone gets excited because it’s an area that has historically been ignored, and then end up disappointed realising it’s the bare minimum a company could do because, surprise, there’s not much competition in the arena of women’s health research, and so there’s no motivation to find the best possible product.
We've seen it with our contraceptive options, most notably the contraceptive pill - which was lauded as revolutionary when it went on the market (and indeed it was life changing for millions of women) - but as The Debrief's #MadAboutThePill campaign discovered - may have uninterrogated links with anxiety and depression. Then there's the hormonal IUD, sold as a reduced-hormone fix for your heavy periods and bleeding, something you don't have to remember to take everyday that COULD EVEN stop your periods altogether. Not only that, it is used to help manage endometriosis, PCOS and painful periods. But studies also show that it may increase your risk of ovarian cysts, ectopic pregnancy and - in my case and many other women - actually leaves you in debilitating pain for months on end while your body adjusts to having a foreign object inside it.
Yet, we still get excited about any new treatment for women's health issues. Because, while the side effects associated with Orilissa demonstrate that it’s far from perfect, the fact that it exists at all is (depressingly) progress. It at least is starting the process to find and explore better drugs and give women more options in an area where there is so little investment. Andrew Horne, a professor of gynaecology and reproductive health at the University of Edinburgh agrees that this is at least a promising progression in the field of treating endometriosis.
‘It’s exciting because it shows that drug companies are investing in new treatments,’ he said, ‘it’s maybe not going to be the treatment everyone will be able to use but at least it’s an option for some women.’
And in fact, he has more hope that women in the UK could benefit from the drug, although (as usual) we might have to get our hands dirty ourselves. ‘Often when drugs are FDA approved it precipitates a similar process for approval within the UK’, he continued, ‘I’m sure AbbVie would be keen that women in the UK can take advantage of this treatment, obviously that might take a bit of time but women themselves will hopefully start to ask if they can have this option available to them in the UK and that sometimes precipitates these things to move forward too.’
It's an optimistic take, and certainly we should be excited about drug companies investing in new treatments in historically ignored areas of research. However, when those treatments come with adverse side-effects and inadequate options, it's just another reason to fight for more research. If we're getting excited about pills that force us into early menopause, thin our bones and possibly make us suicidal, we still have a long way to go.
Click through to read the #MadAboutThePill statistics on how the contraceptive pill can impact your body...
Debrief Mad About The Pill Stats
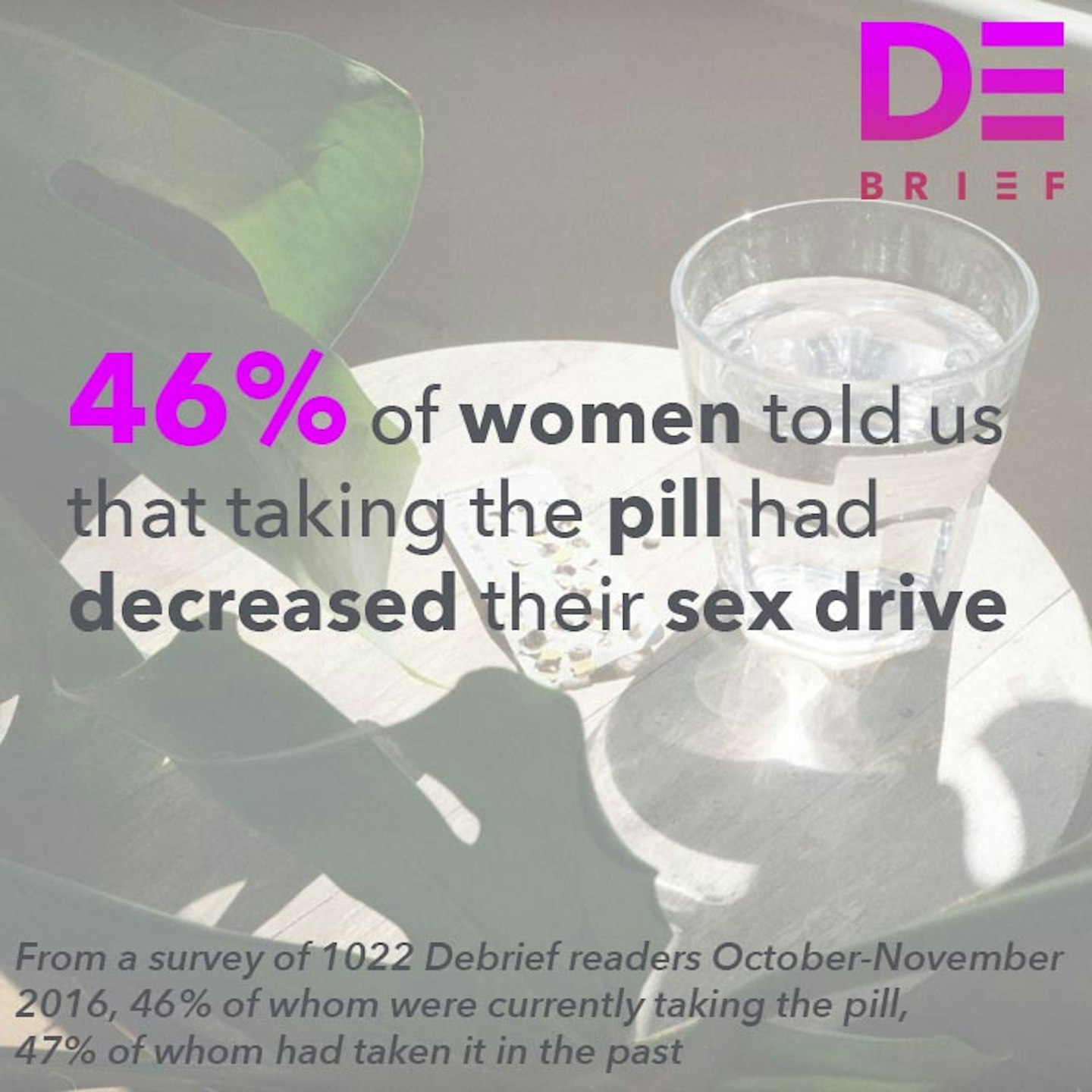 1 of 9
1 of 9Debrief Mad About The Pill Stats
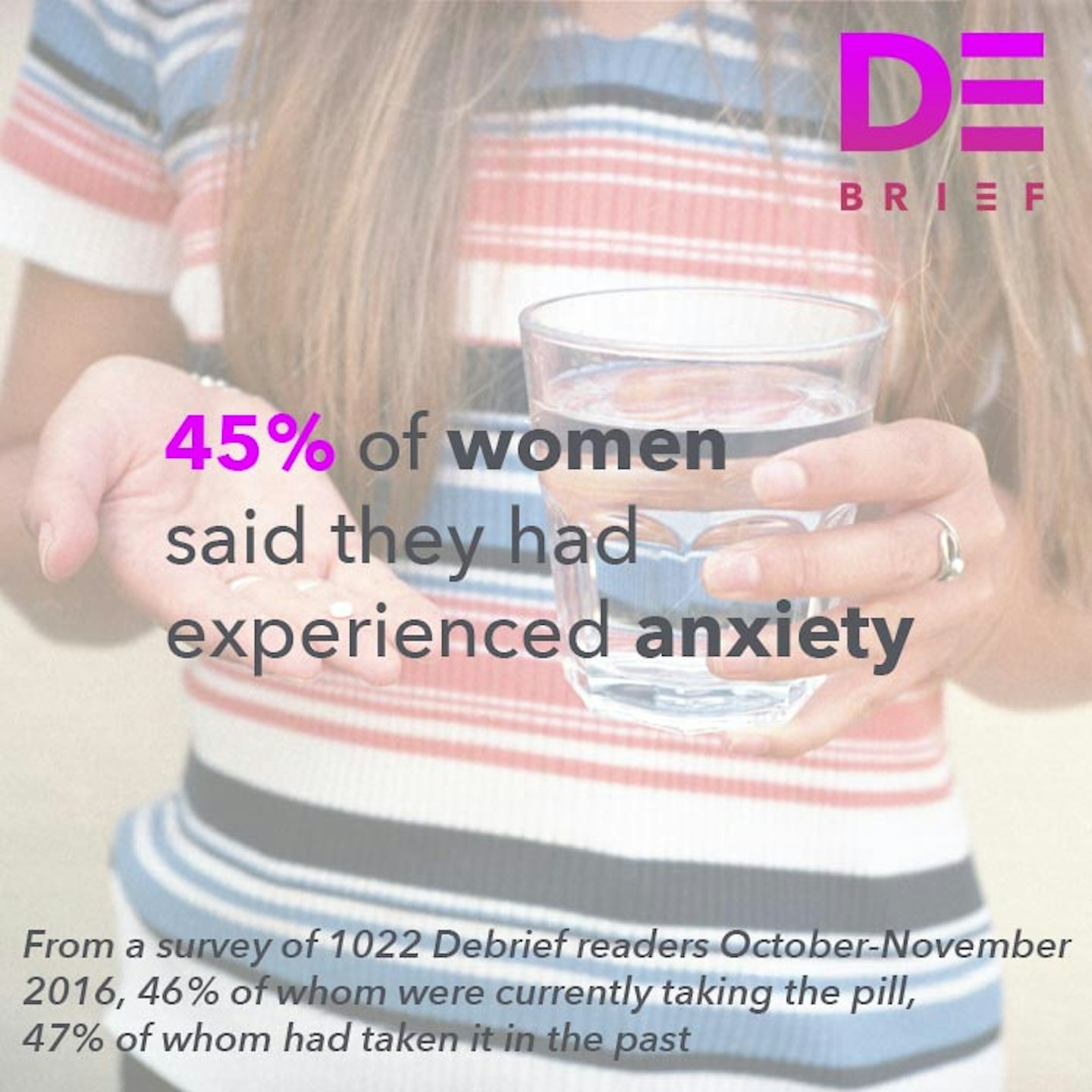 2 of 9
2 of 9Debrief Mad About The Pill Stats
 3 of 9
3 of 9Debrief Mad About The Pill Stats
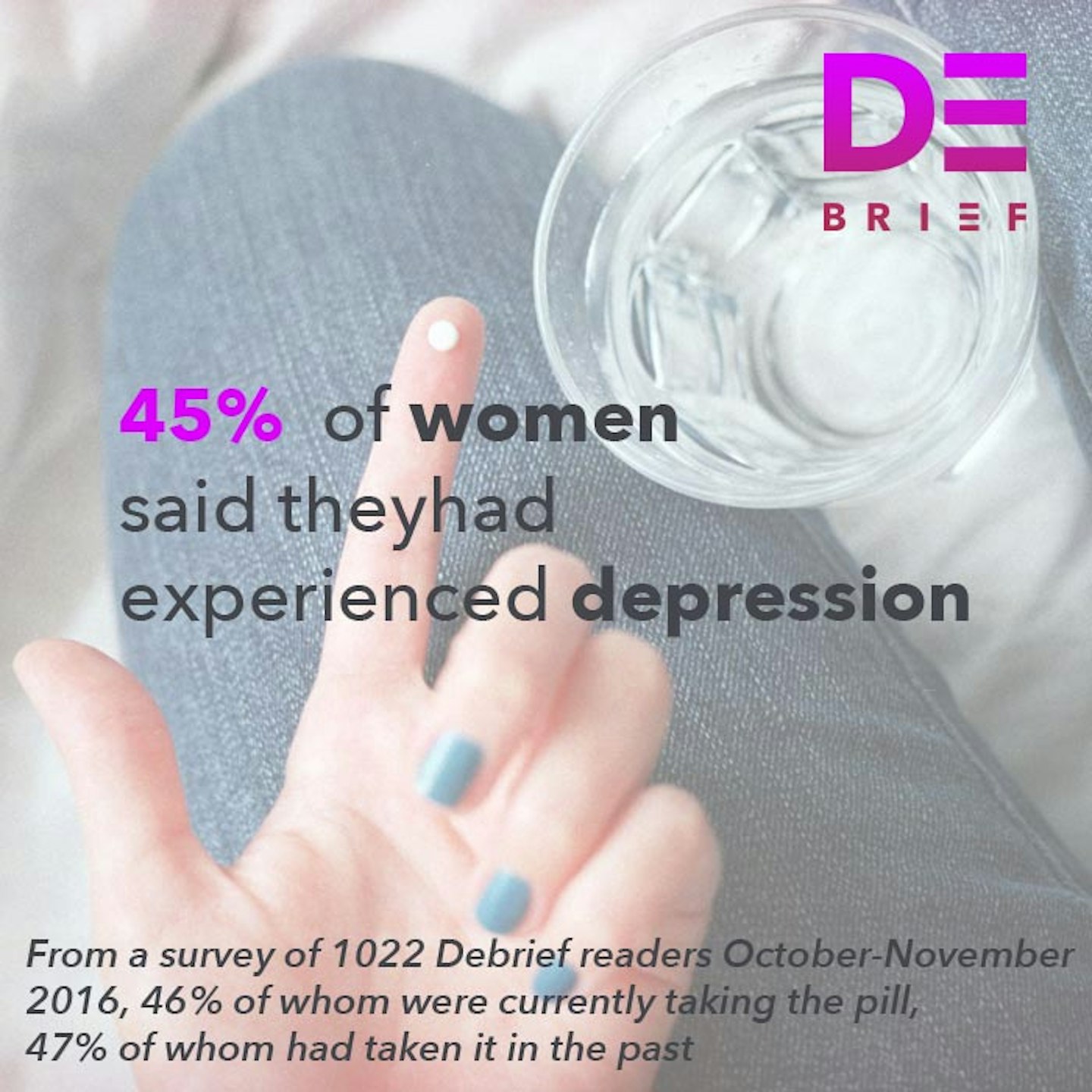 4 of 9
4 of 9Debrief Mad About The Pill Stats
 5 of 9
5 of 9Debrief Mad About The Pill Stats
 6 of 9
6 of 9Debrief Mad About The Pill Stats
 7 of 9
7 of 9Debrief Mad About The Pill Stats
 8 of 9
8 of 9Debrief Mad About The Pill Stats
 9 of 9
9 of 9